
Handy Links
SLAC News Center
SLAC Today
- Subscribe
- Archives: Feb 2006-May 20, 2011
- Archives: May 23, 2011 and later
- Submit Feedback or Story Ideas
- About SLAC Today
SLAC News
Lab News
- Interactions
- Lightsources.org
- ILC NewsLine
- Int'l Science Grid This Week
- Fermilab Today
- Berkeley Lab News
- @brookhaven TODAY
- DOE Pulse
- CERN Courier
- DESY inForm
- US / LHC
SLAC Links
- Emergency
- Safety
- Policy Repository
- Site Entry Form

- Site Maps
- M & O Review
- Computing Status & Calendar
- SLAC Colloquium
- SLACspeak
- SLACspace
- SLAC Logo
- Café Menu
- Flea Market
- Web E-mail
- Marguerite Shuttle
- Discount Commuter Passes
-
Award Reporting Form
- SPIRES
- SciDoc
- Activity Groups
- Library
Stanford
Around the Bay
Physicists Discover a New Structure of Cationic Lipid-DNA Complexes for Gene Therapy
 Gene therapy is one of the most promising future approaches to fighting disease on the molecular level. By inserting genes into an individual's cells and tissues with a DNA carrier (also known as a vector), this novel medical approach seeks to cure cancers, inherited diseases, cardiovascular diseases and many others. Numerous studies have been conducted worldwide and several clinical trials in this field are currently ongoing, in the effort to develop and fundamentally understand gene delivery vectors.
Gene therapy is one of the most promising future approaches to fighting disease on the molecular level. By inserting genes into an individual's cells and tissues with a DNA carrier (also known as a vector), this novel medical approach seeks to cure cancers, inherited diseases, cardiovascular diseases and many others. Numerous studies have been conducted worldwide and several clinical trials in this field are currently ongoing, in the effort to develop and fundamentally understand gene delivery vectors.
Cationic liposome (CL)-DNA complexes are a prevalent non-viral delivery vector, and extended investigations have been performed in order to understand their formation, structure and mechanism of action. The goal is to explore the correlation between their structure and transfection efficiency, thus enabling one to design a vector with desired properties. Working at both our home institution—the University of California, Santa Barbara—and at SSRL, we in Cyrus Safinya's group focus on answering this question.
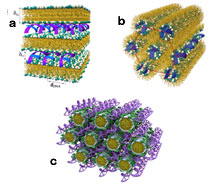
Two structures of CL-DNA complexes have been so far observed: lamellar (LαC) liquid crystal phase and inverted hexagonal (HIIC) phase. Both reported previously by Safinya's group. Most lipids accommodate lamellar phase (Figure a, above), where lipid bilayers alternate with layers of DNA molecules. Lipids with cone-shaped molecular structure prefer the inverted hexagonal phase, where lipids are organized in the inverse cylindrical micelles, which are hexagonally ordered with respect to each other. DNA molecules are localized inside the inverse cylindrical micelles (Figure b, above).
Recently, newly designed multivalent lipids ranging in head group charge from 4+ to 16+ have been synthesized in our lab and investigated as DNA delivery vectors. These dendritic lipids (DLs) allow a controlled study of the relationship between membrane charge density (σ) and transfection efficiency. An earlier report from our group described that TE of different cationic lipids of charge 1+ to 5+ follows a common, bell shaped curve as a function of membrane charge density. To further probe this universal behavior, the dendritic lipids with higher valence were designed in order to reach higher values of σ.
Extensive structural studies using x-ray diffraction have been conducted at SSRL's beamline 4-2. Thanks to huge support by Hiro Tsuruta and his colleagues at SSRL we were able to perform structural investigations, which lead to revealing of the new phase, where cylindrical micelles of dendritic lipids (DL) form a hexagonal lattice which holds together a continuous DNA network, described as HIC. This phase appears in very narrow interval around 25 Mole% of dendritic lipid. The new hexagonal phase (Figure c, above) is highly transfecting in the regime where the TE of lamellar complexes follows a decrease in the above mentioned bell curve (i.e. for higher σ values), demonstrating the importance of understanding both the function and structure of lipid mixtures in the context of improved non-viral gene delivery. A surprising dependence of efficiency on the charge ratio DNA:DL was also observed. Small angle x-ray scattering studies have revealed a rich phase diagram of micelles made from DL/DOPC mixtures. The extreme difference in the molecular shape between DLs and DOPC results in distinct micellar morphologies as a function of DL/DOPC molar ratios. This fact allows us to expect an existence of further so far unknown phases of CL-DNA complexes.
Our studies are only one little piece in a huge mosaic of medical research, however the motivation to understand and develop well functioning gene therapy is immense. We still need to learn much more before we can design an ideal gene delivery vector, but every seemingly little discovery brings us closer to the ultimate goal.
—Alexandra Zidovska
SLAC Today, October 19, 2006
Above image: Structures of CL-DNA complexes: a) lamellar liquid crystal phase LαC, b) inverted hexagonal phase HIIC and c) hexagonal phase HIC