
Handy Links
SLAC News Center
SLAC Today
- Subscribe
- Archives: Feb 2006-May 20, 2011
- Archives: May 23, 2011 and later
- Submit Feedback or Story Ideas
- About SLAC Today
SLAC News
Lab News
- Interactions
- Lightsources.org
- ILC NewsLine
- Int'l Science Grid This Week
- Fermilab Today
- Berkeley Lab News
- @brookhaven TODAY
- DOE Pulse
- CERN Courier
- DESY inForm
- US / LHC
SLAC Links
- Emergency
- Safety
- Policy Repository
- Site Entry Form

- Site Maps
- M & O Review
- Computing Status & Calendar
- SLAC Colloquium
- SLACspeak
- SLACspace
- SLAC Logo
- Café Menu
- Flea Market
- Web E-mail
- Marguerite Shuttle
- Discount Commuter Passes
-
Award Reporting Form
- SPIRES
- SciDoc
- Activity Groups
- Library
Stanford
Around the Bay
A Novel Form of Iron
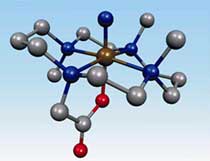 Chemists have synthesized and characterized a new, highly reactive form of iron that promises to deepen our understanding of this important element. Iron is found in abundance in the natural world, and in its ionized form plays a crucial role in virtually all living processes.
Chemists have synthesized and characterized a new, highly reactive form of iron that promises to deepen our understanding of this important element. Iron is found in abundance in the natural world, and in its ionized form plays a crucial role in virtually all living processes.
An iron atom can have as many as eight valence electrons in its outer shell, but most typically iron has five or six of these valence electrons. The new form of iron, designated iron(VI), has only two electrons in its outer shell, making it very electron deficient and thus highly reactive. Only one other form of iron(VI), ferrate, which is tetrahedrally coordinated by four oxygen atoms, is known. Ferrate is a powerful oxidant, but reacts indiscriminately and thus can be difficult to use. The new species of iron(VI) which is octahedrally coordinated, instead forms a bond with nitrogen, which results in different reactivity, and thus holds promise for industrial and biomedical applications.
"We hope that this complex will have practical advantages over other iron compounds, and we might expect that it does based on its structure," explained John F. Berry, an Alexander von Humboldt postdoctoral fellow at Germany's Max-Planck Institute for Bioinorganic Chemistry in Mulheim. Berry is lead author of the paper describing the new compound, which was originally published in Science Magazine's online publication, Science Express.
The research team, which included scientists from the Max-Planck Institute for Bioinorganic Chemistry in Germany and the Stanford Synchrotron Radiation Laboratory, conducted x-ray absorption spectroscopy (XAS) experiments at SSRL's Beam Line 9-3 to characterize the new complex. The XAS studies, combined with other spectroscopic results and theoretical calculations, were key in establishing the electronic and geometric structure of this novel compound.
Read a more technical description of this research in SSRL Highlights.
—Brad Plummer
SLAC Today, August 3, 2006
Above image: The proposed Fe(VI)-nitrido photolysis product.